The Part of the Process That’s Easy to Overlook
Ion exchange is often viewed as a clean solution.
Contaminants are captured onto a resin. Effluent meets specification. The system appears stable and predictable from the outside. For many facilities, that is where the evaluation stops. As long as the discharge is compliant, the process is considered successful.
What tends to receive less attention is what happens next.
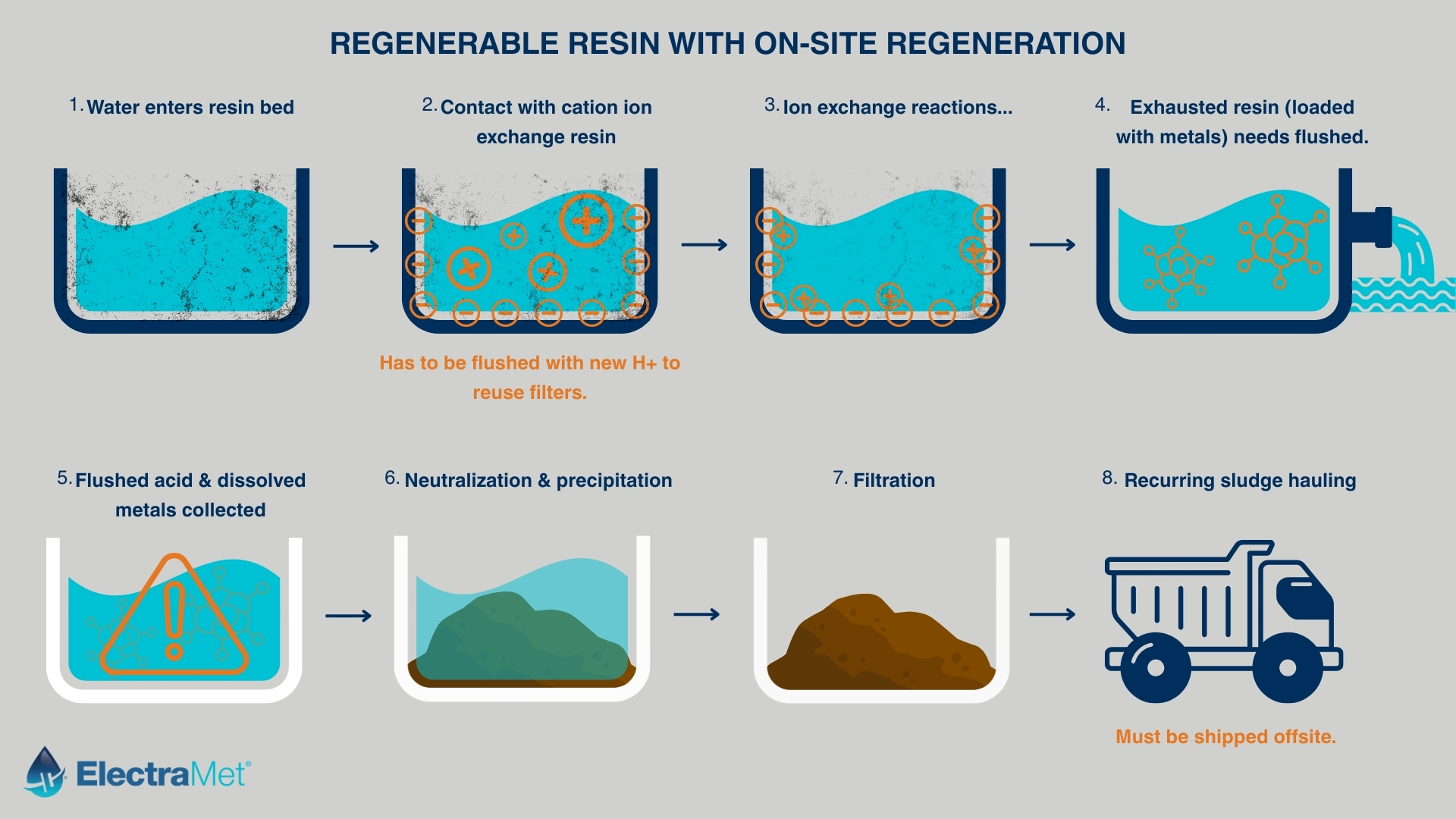
Resins do not remove contaminants permanently. They concentrate them. Over time, those resins reach capacity and must be regenerated. That regeneration step is where the captured material re-enters the system in a different form, and where the overall mass balance of the process becomes more visible.
Regeneration Is a Transfer, Not a Resolution
When ion exchange resins are regenerated, the goal is to restore their capacity for continued use. This is typically achieved by flushing the resin with a chemical solution that strips the captured ions back into liquid form.
At that point, the system has not eliminated the contaminant. It has changed its concentration and location.
What was previously distributed across a larger volume of water is now contained in a smaller, more concentrated stream. This regenerated solution often contains high levels of dissolved metals along with the chemicals used in the regeneration process itself.
From a compliance standpoint, this is expected. From a systems standpoint, it introduces a new layer of complexity.
The Hidden Stream Behind the System
The regeneration step creates a secondary waste stream that is fundamentally different from the original influent.
It is more concentrated.
It is more chemically complex.
And it is often more difficult to handle.
Because this stream is generated intermittently rather than continuously, it can be easy to treat it as an exception rather than part of the core process. In practice, it is a direct result of how the system operates and should be evaluated accordingly.
Facilities typically manage this stream through offsite hauling, further treatment, or a combination of both. Each of these options carries its own cost structure and operational considerations, which are not always visible when the ion exchange system is first implemented.
Where Costs Begin to Accumulate
The economics of ion exchange are often evaluated based on resin performance, replacement intervals, and initial system cost. The downstream implications of regeneration tend to be less prominent in early-stage decision making.
Over time, however, the costs associated with the regeneration stream can become more significant. Concentrated waste often falls into more restrictive classifications, which can increase disposal costs. Handling requirements may become more stringent, and the need for additional treatment steps can introduce further operational burden.
Because these costs are tied to regeneration cycles rather than continuous flow, they may appear episodic. When viewed over a longer horizon, they form a consistent and material part of the overall cost of the system.
The Operational Reality of Managing Regeneration
Beyond cost, regeneration introduces a layer of operational complexity that is not always apparent at the outset.
Chemical handling becomes more involved, particularly when strong acids or bases are used in the regeneration process. Storage and safety considerations must be accounted for, especially when dealing with concentrated metal solutions. Scheduling regeneration cycles to align with production and treatment capacity can also require active management.
None of these challenges are insurmountable, but they shift the system from a passive treatment approach to one that requires ongoing oversight and coordination.
A Different Way to Look at the Same Problem
The key point is not that ion exchange is ineffective. It is that the process does not eliminate contaminants. It concentrates them and creates a secondary stream that must be addressed.
That distinction matters because it changes how the system should be evaluated. Instead of asking whether the primary effluent meets specification, the more complete question is how the entire process handles the material from start to finish.
In some cases, facilities begin to look for approaches that address dissolved contaminants more directly, without creating a regeneration cycle or a concentrated secondary waste stream. Electrochemical systems, for example, can remove metals from solution by converting them into solid forms without the need for periodic chemical regeneration, while oxidant destruction can occur without introducing additional reagents into the system.
The relevance of these approaches depends on the application. The broader takeaway is that different technologies manage the same mass balance in fundamentally different ways.
Where This Becomes a Strategic Consideration
As waste streams become more complex and disposal pathways more constrained, the regeneration step becomes harder to treat as a secondary issue. It is part of the system, both operationally and financially.
Understanding what happens after ion exchange regeneration does not necessarily lead to an immediate change in approach. It does, however, provide a clearer picture of how the system functions as a whole, and where costs and risks may be concentrated.
In that sense, the question is not simply how well a system removes contaminants at the point of discharge, but how effectively it manages those contaminants throughout the entire process.
